The Chemical Context Of Life
Chapter ii. The Chemical Context of Life

- 2.1 Atoms
- 2.ii Chemical Bonding & Intermolecular Forces
- 2.three H2o, Acids & Bases
- 2.4 The Energy of Life
- 2.5 Carbon
Introduction
Elements in various combinations comprise all matter, including living things. Some of the most abundant elements in living organisms include carbon, hydrogen, nitrogen, oxygen, sulfur, and phosphorus. These course the nucleic acids, proteins, carbohydrates, and lipids that are the fundamental components of living matter. Biologists must empathize these important edifice blocks and the unique structures of the atoms that make up molecules, assuasive for the germination of cells, tissues, organ systems, and entire organisms.
All biological processes follow the laws of physics and chemistry; and so in order to understand how biological systems work, it is important to understand the underlying physics and chemistry. For case, the flow of blood inside the circulatory arrangement follows the laws of physics that regulate fluid menses. The breakdown of the large, circuitous molecules of nutrient into smaller molecules—and the conversion of these to release energy to be stored in adenosine triphosphate (ATP)—is a series of chemical reactions that follow chemical laws. The properties of water and the germination of hydrogen bonds are key to agreement living processes. Recognizing the backdrop of acids and bases is important, for instance, to our understanding of the digestive process. Therefore, the fundamentals of physics and chemistry are of import for gaining insight into biological processes.
2.1 | Atoms
By the end of this department, yous will be able to:
- Ascertain thing and elements.
- Draw the interrelationship between protons, neutrons, and electrons.
- Use diminutive number to determine electron configuration.
At its virtually fundamental level, life is made upward of affair: any substance that occupies space and has mass. Elements are unique forms of matter with specific chemical and physical properties that cannot be broken downwardly into smaller substances by ordinary chemical reactions. There are 118 elements, simply but 92 occur naturally. The remaining elements are synthesized in laboratories and are unstable.
Each element is designated past its chemical symbol, which is a single capital alphabetic character or, when the first letter is already used for another element, a combination of two letters. Some elements follow the English term for the element, such every bit C for carbon and Ca for calcium. Other elements' chemical symbols derive from their Latin names; for instance, the symbol for sodium is Na, referring to natrium, the Latin word for sodium.
The four most mutual elements in all living organisms are oxygen (O), carbon (C), hydrogen (H), and nitrogen (Due north). These elements comprise 96% of living organisms. In the non-living world, elements are found in different proportions, and some elements common to living organisms are relatively rare on the earth as a whole, as shown in Table 2.1. In spite of their differences in abundance, all elements and the chemical reactions between them obey the same chemical and physical laws regardless of whether they are a part of the living or not-living earth.
Table two.1. Percent of elements in living organisms vs. the non-living world.
| Element | Life (Humans) | Atmosphere | Globe's Crust |
| Oxygen (O) | 65% | 21% | 46% |
| Carbon (C) | eighteen% | trace | trace |
| Hydrogen (H) | ten% | trace | 0.i% |
| Nitrogen (N) | 3% | 78% | trace |
ii.one.1 The Structure of the Atom
An atom is the smallest unit of measurement of matter that retains all of the chemic properties of an element. For instance, ane gilt atom has all of the properties of gold, such as that it is a solid metal at room temperature. Gilt atoms cannot be broken down into anything smaller while still retaining the backdrop of gold.
An atom is composed of two regions: the nucleus, which is in the center of the cantlet and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus (Figure 2.2). Atoms contain subatomic particles, the largest of which are protons, electrons, and neutrons.
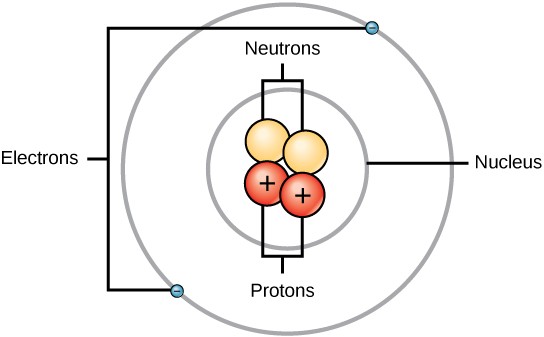
P roton s are positively charged, electrons are negatively charged, and neutron s are uncharged (Table two.2). Each electron has a negative accuse equal to the positive charge of a proton. In uncharged, neutral atoms, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. In these atoms, the positive and negative charges cancel each other out, leading to an cantlet with no net charge.
Protons and neutrons have approximately the same mass, about one.67 × 10-24 grams. Scientists arbitrarily define this amount of mass as 1 atomic mass unit (amu) or 1 Dalton (Da) (Table 2.ii). Electrons are much smaller in mass than protons, weighing only ix.xi × 10-28 grams, or about one/1800 of an atomic mass unit. Hence, they practice not contribute much to an chemical element's overall atomic mass. Therefore, when considering diminutive mass, it is customary to ignore the mass of any electrons and summate the cantlet's mass based on the number of protons and neutrons alone.
Accounting for the sizes of protons, neutrons, and electrons, nigh of the volume of an atom—greater than 99 pct—is, in fact, empty infinite. With all this empty space, 1 might ask why so-called solid objects do not simply pass through one another. The reason they do non is that the electrons that surround all atoms are negatively charged and negative charges repel each other.
Tabular array 2.2. Backdrop of subatomic particles.
| Accuse | Mass (amu) | Location | |
| Proton | +1 | 1 | nucleus |
| Neutron | 0 | one | nucleus |
| Electron | –1 | 0 | orbitals |
2.1.2 Diminutive Number, Mass Number, Isotopes, and Atomic Weight
Atoms of each element contain a characteristic number of protons. The number of protons determines an element's atomic number and is used to distinguish 1 element from another.
The number of neutrons in the atoms of a given element is variable. For example, the chemical element carbon (C) has atomic number vi. Therefore, all neutral carbon atoms have 6 protons and 6 electrons. Even so, some carbon atoms have six neutrons, some have 7 neutrons, and some accept 8 neutrons. Together, the number of protons plus the number of neutrons determines an atom'southward mass number. Note that the minor contribution of mass from electrons is disregarded when computing the mass number.
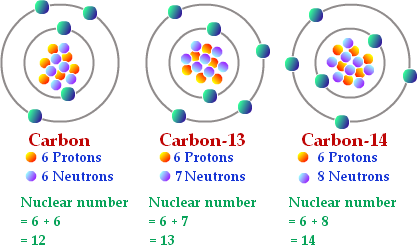
Isotopes are atoms that have the same number of protons only a unlike number of neutrons. Carbon atoms that take 6 neutrons have a mass number of 12 amu, and are referred to as Carbon-12 or 12C. Carbon atoms with 7 neutrons take a mass number of 13 amu, and are referred to as Carbon-13 or 13C. Carbon atoms that accept 8 neutrons have a mass number of xiv amu, and are referred to as Carbon-14 or xivC. These stand for three naturally occurring isotopes of carbon (Effigy 2.iii).
Since an chemical element's isotopes accept different mass numbers, scientists also determine the atomic weight, which is the calculated mean of the mass number for the naturally occurring isotopes of an chemical element on earth. Frequently, the resulting number is not a whole number. For example, the atomic mass of chlorine (Cl) is 35.45 because chlorine is equanimous of several isotopes, some (the majority) with diminutive mass 35 (17 protons and eighteen neutrons) and some with atomic mass 37 (17 protons and 20 neutrons). The atomic mass of carbon is 12.011 because the peachy bulk of carbon on earth is Carbon-12.
Some isotopes may emit neutrons, protons, and electrons in lodge to go more stable. These are radioactive isotopes, or radioisotopes. Radioactive decay describes the loss of energy and/or mass that occurs when an unstable cantlet'due south nucleus releases radiation. Carbon-fourteen is an case of a radioisotope (Figure 2.4).

Carbon DatingCarbon-fourteen (14C) is a naturally occurring radioisotope. In a living organism, the relative amount of 14C is equal to the concentration of 14C in the atmosphere. When an organism dies, the ratio between 14C and 12C will decrease as 14C decays.After approximately v,730 years, half of the starting concentration of fourteenC rust-covered. The time it takes for half of the original concentration of an isotope to decay to its more than stable form is called its half-life. The number of half-lives since an object such equally bones or wood was alive tin can exist determined by comparing the ratio of the fourteenC concentration in the object to the amount of 14C detected in the temper. The age of the fabric can be calculated with accurateness if it is not much older than about 50,000 years (Figure ii.4).

2.1.3 The Periodic Tabular array
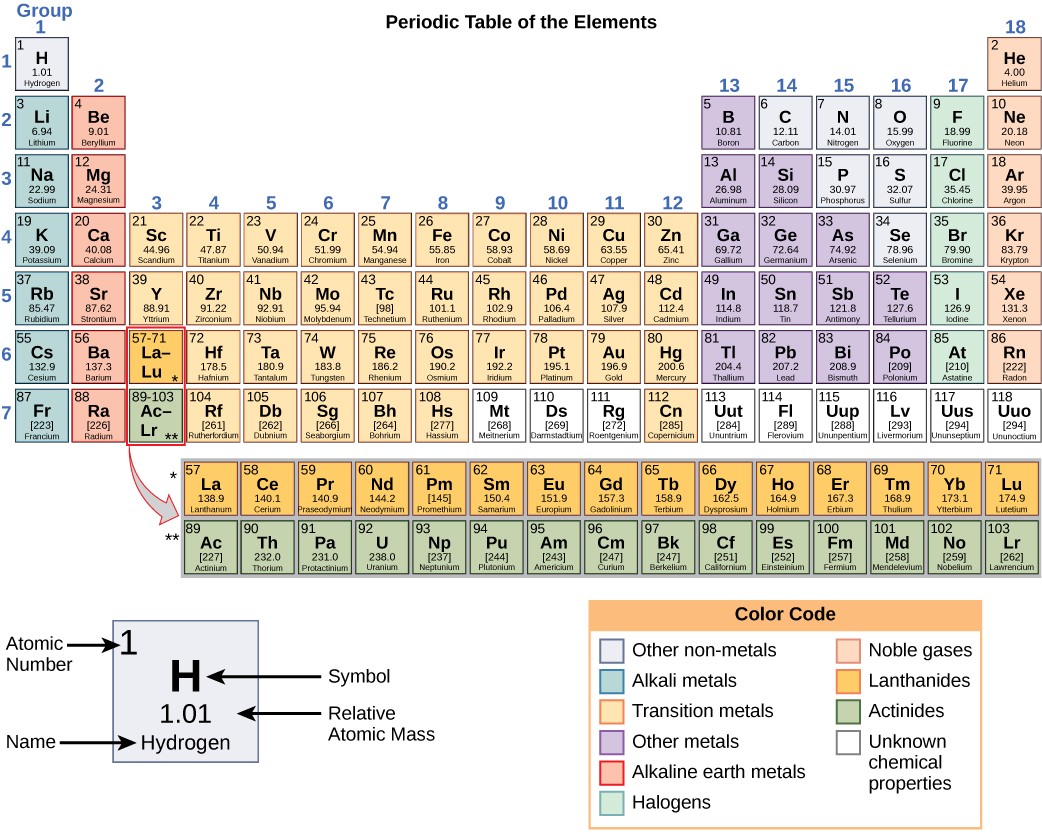
The different elements are organized and displayed in the periodic table. Devised past Russian chemist Dmitri Mendeleev (1834–1907) in 1869, the table groups elements that share sure chemical properties. The properties of elements are responsible for their concrete state at room temperature: they may be gases, solids, or liquids. Elements also have specific chemical reactivity, the ability to chemically bond with each other.
In the periodic tabular array, shown in Figure two.v, the elements are organized and displayed according to their diminutive number and are arranged in a series of rows and columns based on shared chemical and physical properties. Each square on the table gives the name, chemical symbol, diminutive weight, and atomic mass for ane element. For example, the first foursquare contains hydrogen, its symbol (H), its diminutive number of (1), and its atomic mass (1.01).
ii.i.4 Electron Shells and the Bohr Model
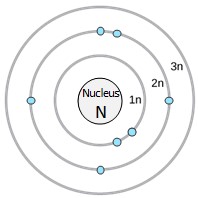
An early model of the atom was developed in 1913 by Danish scientist Niels Bohr (1885–1962). The Bohr model shows the atom with a central nucleus and the electrons in circular electron shells at specific distances from the nucleus. The closer an energy shell is the nucleus, the lower the energy of the electrons that occupy that shell. The showtime (1n) electron vanquish can agree two electrons, while the 2d (2n) and 3rd (3n) shells can concord viii electrons each (Figure 2.vi).
Electron Orbitals
Although useful to explain the reactivity and chemical bonding of sure elements, the Bohr model of the atom does not accurately reverberate how electrons are spatially distributed surrounding the nucleus. They do not circle the nucleus like the earth orbits the sun, simply are found in three-dimensional electron orbitals. Mathematical equations tin predict within a certain level of probability where an electron might exist at any given time. The area where an electron is most likely to be found is called its orbital. Each electron orbital can hold just two electrons.
Electrons make full orbitals in a consistent order: they first fill the orbitals closest to the nucleus, so they continue to fill orbitals of increasing free energy further from the nucleus. If there are multiple orbitals of equal energy, one electron will be added to each orbital earlier a 2d electron is added to whatsoever of the orbitals. For case, the second energy level in the nitrogen atom shown here has one electron in each of three orbitals and ii electrons in the fourth orbital (Figure 2.half-dozen).
The innermost shell has a single orbital, for a maximum of two electrons only the next two electron shells can each accept four orbitals, for a maximum of eight electrons. The octet rule states that, with the exception of the innermost shell, atoms are most stable when they accept viii electrons in their valence shell, the outermost electron shell. Examples of some neutral atoms and their electron configurations are shown in Figure 2.7.
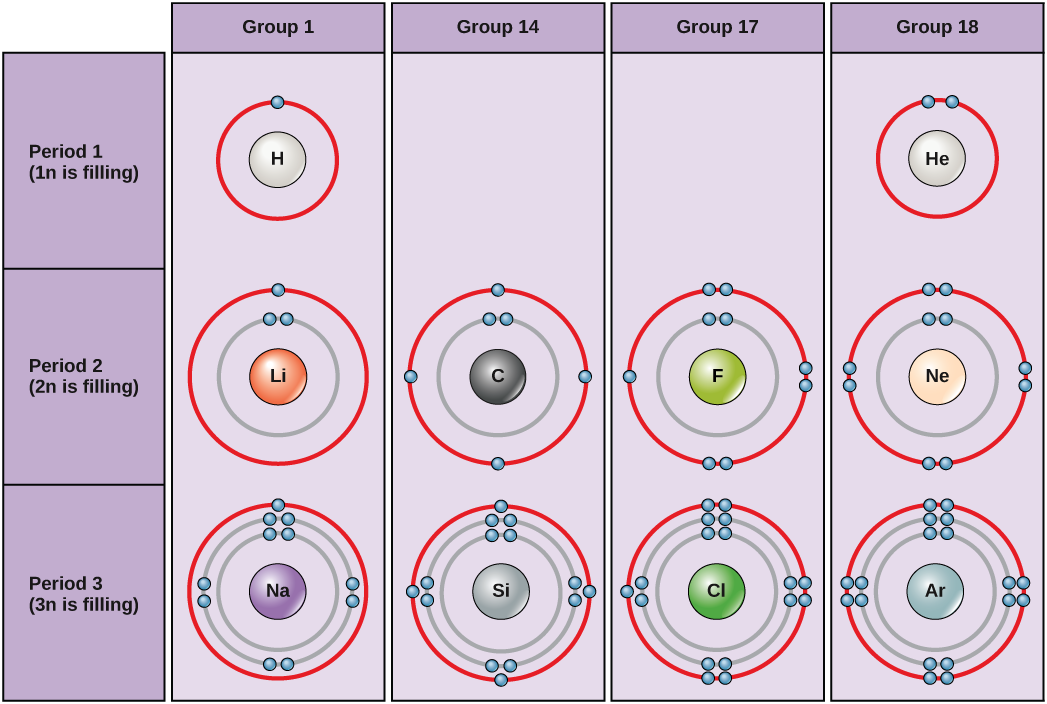
Describe Bohr'southward diagrams for oxygen and magnesium atoms.
- How many electrons do oxygen atoms need to proceeds in club to reach a stable electron configuration?
- How many electrons do magnesium atoms need to lose to attain a stable configuration?
The periodic tabular array is arranged in columns and rows based on the number of electrons and where these electrons are located. Notation that elements in the far right column of the periodic tabular array (Figure 2.five) all have filled valence shells. These atoms are highly stable, making it unnecessary for them to share electrons with other atoms. They are therefore non-reactive and are chosen inert gases (or noble gases).
In full general, atoms with 4-7 electrons in their valence vanquish will either gain electrons to become negatively charged ions, or will share electrons with other atoms to form covalently bonded molecules. Atoms with 1-three electrons in their valence beat out will tend to donate their electrons to other atoms until they have a full outer vanquish. As a issue of losing negatively charged electrons, they get positively charged ions.
2.2 | Chemical Bonding & Intermolecular Forces
Past the end of this department, you will be able to:
- Compare the ways in which electrons can be donated or shared betwixt atoms.
- Explain the means in which naturally occurring elements combine to create molecules.
- Place intermolecular forces that hold molecules together.
2.ii.1 Chemic Reactions and Molecules
The octet dominion drives the chemical behavior of atoms. Atoms will chemically react and bond to each other form molecules, which are simply two or more atoms chemically bonded together. A compound is a blazon of molecule that contains two or more dissimilar types of atoms. In short, atoms form chemical bonds with other atoms, thereby obtaining the electrons they need to achieve a stable electron configuration. The familiar water molecule, H2O, consists of 2 hydrogen atoms and 1 oxygen atom bonded together (Figure 2.8). Atoms tin form molecules by altruistic, accepting, or sharing electrons to fill their outer shells.
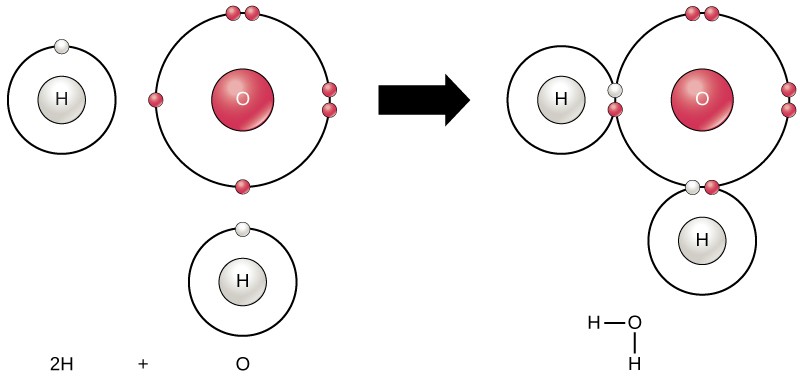
Chemical reactions occur when two or more than atoms bond together to course molecules or when bonded atoms are broken apart. The substances used in the beginning of a chemical reaction are called the reactants and the substances constitute at the terminate of the reaction are known equally the products . An arrow is typically fatigued betwixt the reactants and products to indicate the direction of the chemical reaction. Most chemical reactions can become in either direction. For the creation of the h2o molecule shown above, the chemical equation would exist:
two Htwo + Otwo → 2 H2O
This is an example of a balanced chemical equation, wherein the number of atoms of each chemical element is the same on each side of the equation.
2.2.2. Ions and Ionic Bonds
Some atoms are more stable when they gain or lose an electrons and class ions. This fills their outermost electron beat and makes them more stable. Considering the number of electrons does not equal the number of protons, each ion has a cyberspace charge. Cations are positive ions that are formed by losing electrons. Anions are negative ions that are formed past gaining electrons.
Certain ions, such as sodium, potassium, and calcium, are referred to in physiology as electrolytes. These ions are necessary for nerve impulse conduction, muscle contractions and h2o residuum. Many sports drinks and dietary supplements provide these ions to supplant those lost from the body via sweating during exercise.
Movement of electrons from ane atom or molecule to some other is referred to as electron transfer or as a redox reaction. As Figure 2.9 illustrates, sodium (Na) merely has one electron in its outer electron shell. If sodium loses an electron, information technology now has eleven protons and only x electrons, making it a sodium cation with an overall charge of +one. Chlorine (Cl) has 7 electrons in its outer shell. If it gains an electron, it now has 17 protons and 18 electrons, making it a chloride anion with an overall accuse of -1. Both ions at present satisfy the octet rule and take consummate outermost shells.
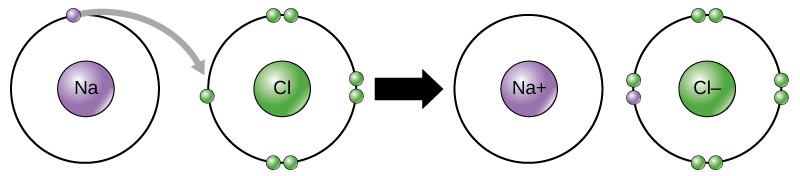
An i onic bond is the electrical allure that forms between ions with reverse charges. For example, positively charged sodium ions and negatively charged chloride ions bond together to make crystals of sodium chloride, or common salt, creating a crystalline molecule with zero internet accuse.
2.2.three Covalent Bonds
Some other way the octet rule can be satisfied is by the sharing of electrons between atoms to form covalent bonds. One, ii, or three pairs of electrons may be shared, making single, double, and triple bonds, respectively.
The germination of water molecules provides an instance of covalent bonding (Effigy two.8). To completely fill up the outer vanquish of oxygen, which has half dozen electrons in its outer shell, two electrons (one from each hydrogen atom) are needed. The electrons are shared between the two elements to fill the outer beat of each, making both elements more stable.
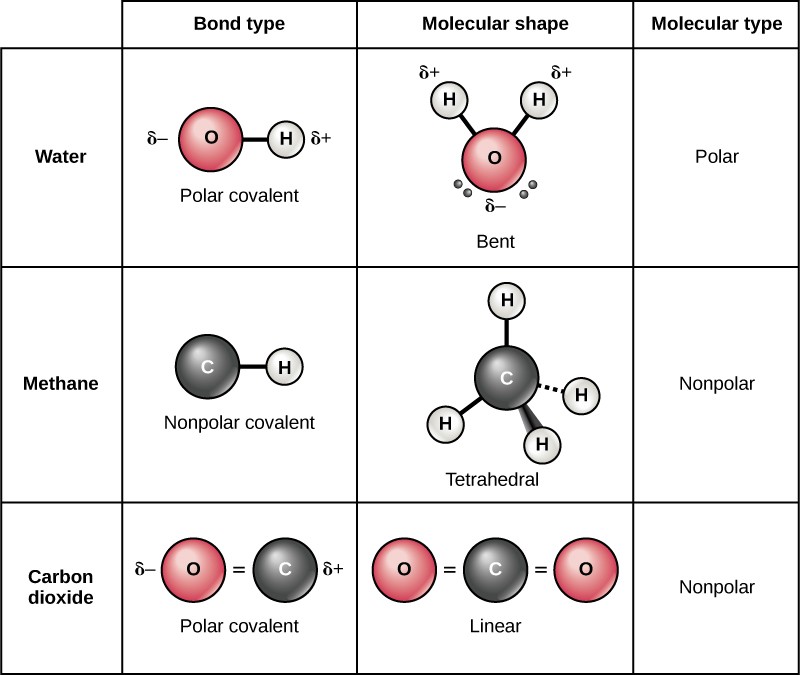
Polar Covalent Bonds
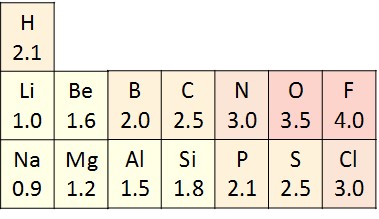
Although atoms share electrons in covalent bonds, they do not always share the electrons equally. Atoms take unlike electronegativities, or attraction for electrons (Figure ii. 1 0). When a covalent bond is formed between two atoms with different electronegativities, the shared electrons volition spend more fourth dimension around the nucleus of the atom with the college electronegativity and less time around the nucleus with lower electronegativity. Since electrons are negatively charged, the atom that gets more time with the electron acquires a slightly negative charge (δ–). The atom with lower electronegativity gets less time with the electron and acquires a slightly positive charge (δ+).
The type of covalent bond that forms between two atoms with unlike electronegativities is called a polar covalent bond. Molecules with polar covalent bonds are called polar molecules, due to the separation of charges beyond the molecule. For example, water is a polar molecule, since oxygen has an electronegativity of 3.5 and hydrogen has an electronegativity of two.1. The oxygen atom in a h2o molecule attracts the shared electrons more and acquires a fractional negative charge, while the hydrogen atoms attract the shared electrons less and acquire a partial positive charge (Effigy 2.i 1). Many of the of import backdrop of water result from its polarity.
Nonpolar Covalent Bonds
Nonpolar covalent bonds form betwixt two atoms of the same element or between different elements that share electrons every bit. For case, molecular oxygen (O2) is nonpolar because the electrons will be equally distributed between the two oxygen atoms.
Some other example of a nonpolar covalent bond is methane (CH4), also shown in Figure 2.1 1. Carbon and hydrogen have like electronegativity values. Therefore, these elements share electrons equally, creating a nonpolar covalent molecule. Some molecules are nonpolar due to symmetry, as seen in the carbon dioxide molecule in Figure ii.11.
2.2.four Hydrogen Bonds and Van Der Waals Interactions
As described above, covalent and ionic bonds occur betwixt atoms to grade molecules. Other types of interactions occur between molecules and are therefore chosen intermolecular forces. Two examples of weak attractions that occur frequently between molecules are hydrogen bonds and van der Waals interactions. Without these two types of attractions, life as we know it would not exist.
Hydrogen bonds are weak interactions between 2 polar molecules or between partially charged parts of molecules. The δ+ of the hydrogen from one molecule is attracted to the δ– charge on the more electronegative atoms (usually oxygen or nitrogen) of another molecule. Hydrogen bonds can too occur between unlike parts of the same molecule. Individual hydrogen bonds are weak and easily broken; however, they occur in very large numbers in h2o and in organic polymers, forming very strong cumulative interactions. Hydrogen bonds between water molecules provide many of the critical, life-sustaining backdrop of water and likewise stabilize the structures of proteins and Deoxyribonucleic acid.
Like hydrogen bonds, van der Waals interactions are weak attractions or interactions between molecules. Van der Waals attractions can occur between whatever two or more molecules and are dependent on slight fluctuations of the electron densities, around an atom. For these attractions to happen, the molecules need to be very close to one another. Although weaker than hydrogen bonds, van der Waals interactions are also additive and tin exist quite strong in keen numbers.
Pharmaceutical Chemist
Pharmaceutical chemists are responsible for developing new drugs and for trying to determine the style of action of drugs. Drugs tin can be found in nature or can be synthesized in the laboratory. In many cases, potential drugs found in nature are changed chemically in the laboratory to make them safer and more than effective.
After the initial discovery or synthesis of a drug, the chemist develops the drug, perhaps by chemically altering information technology, testing to see if it is toxic, and designing methods for large-scale production. Adjacent, the process of getting the drug approved for human use by the Food and Drug Administration (FDA) begins. This involves a series of large-scale experiments using human subjects to make sure the drug is safe and effective. Approval often takes several years and requires the participation of physicians and chemists.
An example of a drug that was originally discovered in a living organism is Paclitaxel (Taxol), an anti-cancer drug used to treat breast cancer. This drug was discovered in the bark of the pacific yew tree. Another case is aspirin, which was originally isolated from willow tree bark. Both of these drugs are at present produced synthetically. Finding drugs oft means testing hundreds of samples of plants, fungi, and other forms of life to encounter if whatsoever biologically active compounds are institute inside them. Sometimes, traditional medicine can give modern medicine clues to where an agile compound can be found. For example, the use of willow bark to brand medicine has been known for thousands of years, dating back to ancient Egypt. It was non until the tardily 1800s, still, that the aspirin molecule, known as acetylsalicylic acid, was purified and marketed for human use.
Occasionally, drugs adult for ane utilize are found to have unforeseen effects that allow these drugs to be used in other, unrelated means. For instance, the drug minoxidil (Rogaine) was originally adult to treat high blood pressure. When tested on humans, information technology was noticed that individuals taking the drug would grow new hair. Eventually the drug was marketed to men and women with baldness to restore lost hair.
The career of the pharmaceutical pharmacist may involve detective work, experimentation, and drug development, all with the goal of making man beings healthier.
2.3 | H2o, Acids & Bases
By the end of this section, you will be able to:
- Describe the properties of water that are critical to maintaining life.
- Explain why water is an excellent solvent.
- Provide examples of water's cohesive and agglutinative properties.
- Hash out the office of acids, bases, and buffers in homeostasis.
Why exercise scientists spend fourth dimension looking for water on other planets? It is considering water is essential to life as we know it. Water is ane of the more than abundant molecules and the one nearly critical to life on Earth. Approximately 60–70 percent of the man body is made up of water. Without it, life as nosotros know it simply would not exist.
The polarity of the water molecule and its resulting hydrogen bonding make h2o a unique substance with special properties that are intimately tied to the processes of life. Life originally evolved in a watery environment, and most of an organism'south cellular chemistry and metabolism occur inside the watery contents of the cell's cytoplasm. Special backdrop of water include its high heat chapters and estrus of vaporization, its ability to dissolve polar molecules, its cohesive and adhesive properties, and its dissociation into ions that leads to the generation of pH. Agreement these characteristics of water helps to elucidate its importance in maintaining life.
2.3.1 The Properties of Water
The Polarity of H2o

One of h2o's important properties is that it is equanimous of polar molecules. While there is no cyberspace charge to a water molecule, the slight positive charges on the hydrogen atoms and the slight negative charges on the oxygen atoms contribute to water's properties of attraction.
As a result of their polarity, water molecules form hydrogen bonds with each other. Water also attracts, or is attracted to, other polar molecules and ions. A polar substance that interacts readily with or dissolves in water is referred to as hydrophilic (hydro- = "water"; -philic = "loving"). In dissimilarity, non-polar molecules, such every bit oils and fats, do not interact well with water, and split from information technology rather than dissolving in information technology (Effigy 2.1 2). These nonpolar compounds are called hydrophobic (hydro- = "water"; -phobic = "fearing").
Water's States: Gas, Liquid, and Solid
Its many hydrogen bonds crusade water to have some unique chemical characteristics compared to other liquids. Since living things take a high water content, understanding these chemical features is key to understanding life. In liquid water, hydrogen bonds constantly form and break as the water molecules slide past each other. The bonds break due to the motion (kinetic free energy) of the water molecules due to the heat contained in the organization. Equally water is boiled, the higher kinetic energy of the h2o molecules causes the hydrogen bonds to break completely and allows h2o molecules to escape into the air as gas (steam or water vapor).
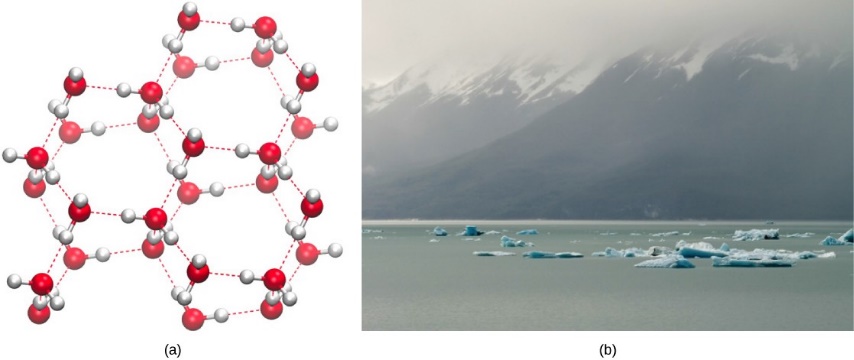
On the other manus, when water freezes, the water molecules form a crystalline structure that makes ice less dumbo than liquid water. Water is less dense every bit a solid because as h2o freezes the water molecules are pushed farther autonomously by the hydrogen bonds. In most other substances, molecules pack more tightly as they freeze, making the solid more dense than the liquid. The lower density of water ice causes it to float on liquid water (Figure ii.13). In lakes and ponds, ice will class on the surface of the water creating an insulating bulwark that protects the living organisms in the swimming from freezing. Without this layer of insulating ice, organisms living in the pond would freeze in the solid block of ice and could non survive.
Water'southward High Heat Capacity
Water has the highest specific heat capacity of whatsoever liquid. Specific rut is defined equally the corporeality of heat one gram of a substance must absorb or lose to change its temperature by one degree Celsius. Because of all of the hydrogen bonds betwixt h2o molecules, it takes water a long fourth dimension to heat and long time to cool. In fact, the specific estrus capacity of h2o is about five times more than that of sand. This explains why the country cools faster than the bounding main. Due to its high heat chapters, water is used by warm blooded animals to help maintain an even temperature.
H2o'due south Rut of Vaporization
Water likewise has a loftier oestrus of vaporization, the amount of free energy required to alter ane gram of a liquid substance to a gas. A considerable corporeality of heat free energy is required to accomplish this change in water. Every bit liquid water heats up, hydrogen bonding makes information technology difficult to separate the liquid water molecules from each other, in social club for it to enter its gaseous stage (steam). Equally a issue, water requires much more heat to boil than most other liquids. Eventually, as h2o reaches its humid point, the estrus is able to interruption the hydrogen bonds between the water molecules, and the kinetic energy (motility) between the water molecules allows them to escape from the liquid as a gas.
Even when beneath its boiling bespeak, private water molecules acquire enough free energy from other water molecules that some tin escape, in a process known as evaporation. Since the evaporation of water requires estrus free energy, information technology cools the environs where the evaporation is taking place. In many living organisms, including humans, the evaporation of sweat allows the organism to absurd so that homeostasis of torso temperature can be maintained.
Water's Solvent Properties
Since water is a polar molecule with slightly positive and slightly negative charges, ions and polar molecules can readily dissolve in information technology. Therefore, water is referred to as a solvent, a substance capable of dissolving other molecules. The substance that is dissolved in a liquid is called a solute. The mixture of a solute dissolved in a solvent is called a solution. If the solvent is h2o, the solution is called an aqueous solution.
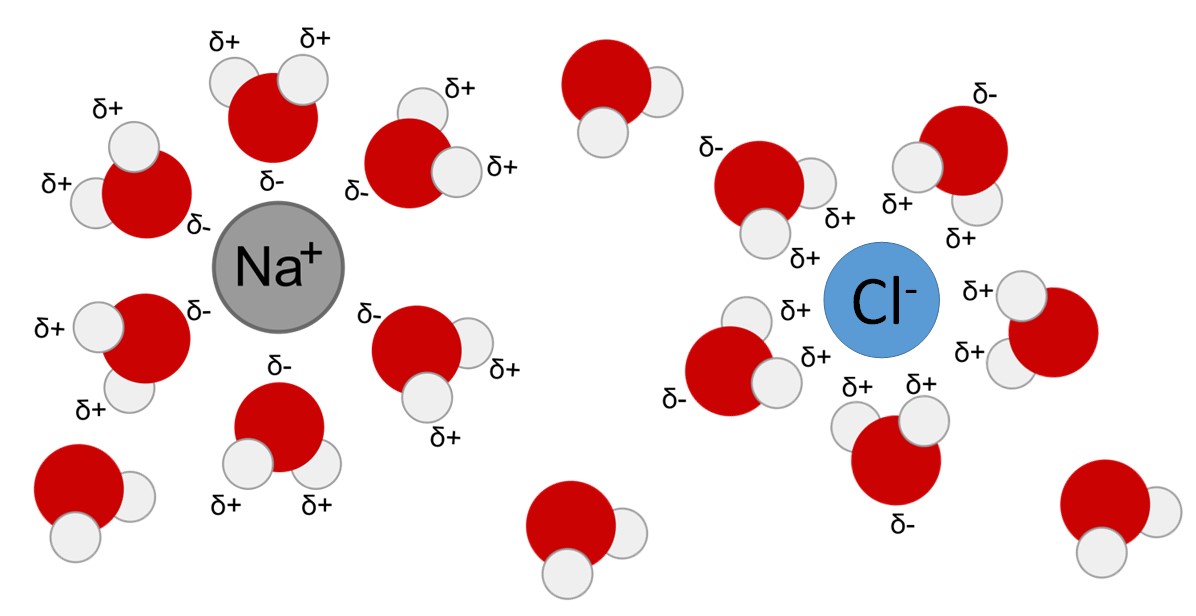
Due to their charge, polar molecules and ions form hydrogen bonds with water and become surrounded by water molecules. The resulting sphere of hydration, or hydration shell, serves to keep the particles separated in the h2o. When ionic compounds are added to water, the individual ions react with the water molecules and their ionic bonds are disrupted in the process of dissociation. For example, when crystals of table table salt (NaCl, or sodium chloride) are added to water, the molecules of NaCl dissociate into Na+ and Cl– ions, and spheres of hydration form around the ions. The positively charged sodium ion is surrounded by the partially negative charge of the water molecule's oxygen. The negatively charged chloride ion is surrounded by the partially positive charge of the hydrogen on the h2o molecule (Figure ii.fourteen).
H2o's Cohesive and Agglutinative Properties
Accept you ever filled a glass of h2o to the very top and and so slowly added a few more than drops? Earlier it overflows, the water forms a dome-similar shape to a higher place the rim of the glass. This h2o tin stay above the glass because of the belongings of cohesion. In cohesion, water molecules are attracted to each other, keeping the molecules together at the h2o-air interface.
Cohesion allows for the development of surface tension, the chapters of a substance to withstand existence ruptured when placed under tension or stress. This is also why h2o forms aerosol when placed on a dry out surface rather than beingness flattened out past gravity. Information technology is fifty-fifty possible to "float" a needle on top of a glass of water or for a water strider to stay afloat on the surface layer of water (Figure 2.15).


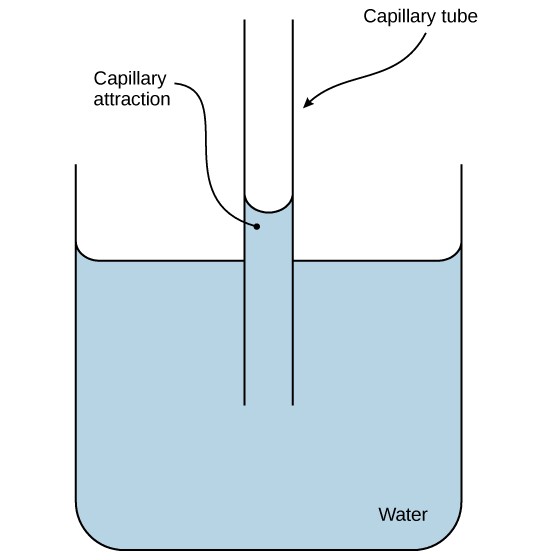
Hydrogen bonding also leads to water's property of adhesion, or the allure of water molecules to other molecules. Adhesion is observed when h2o "climbs" up the tube placed in a glass of water: notice that the h2o appears to be higher on the sides of the tube than in the middle. This is considering the water molecules are attracted to the charged glass walls of the capillary and therefore attach to it. This blazon of adhesion is called capillary action (Figure 2.16).
Cohesion and adhesion are important for the send of water from the roots to the leaves in plants. As water molecules are evaporated from the surface of leaves, they tend to stay connected to water molecules below them, creating a "pull" up the h2o cavalcade. Ultimately, h2o is pulled into the roots, assuasive the plant to receive the dissolved minerals they crave from the soil.
2.iii.ii Acids and Bases
pH is a measure of the concentration of hydrogen ions [H+]in a solution. Hydrogen ions are spontaneously generated in pure water past the dissociation (ionization) of a minor percent of water molecules into equal numbers of hydrogen ions (H+) and hydroxide ions (OH–) ions. While the hydroxide ions are kept in solution past hydrogen bonding with other h2o molecules, the hydrogen ions are immediately attracted to water molecules, forming hydronium ions (H30+). Nevertheless, by convention, scientists refer to hydrogen ions and their concentration as if they were free in this state in liquid water.
H2O ↔ H++OH–
H+ +H2O ↔HthreeO+
The concentration of hydrogen ions dissociating from pure water is 1 × 10-7 moles H+ ions per liter of water. I mole of a substance is equal to 6.02 x 1023 particles of the substance. pH is calculated as the negative of the base x logarithm of the H+ concentration. The logx of 1 × ten-7 is -7.0, and the negative of this number yields a pH of 7.0, which is besides known equally neutral pH. The pH inside cells (half dozen.8) and the pH of human claret (seven.4) are both very close to neutral. Extremes in pH in either direction are commonly considered inhospitable to life.
An acid is a substance that increases the H+ concentration in a solution, commonly past having ane of its hydrogen atoms dissociate. A base provides either OH– or other negatively charged ions that combine with hydrogen ions, reducing their concentration in the solution and thereby raising the pH. In cases where the base releases hydroxide ions, these ions bind to complimentary hydrogen ions, generating new water molecules.
The stronger the acid, the more readily it donates H+. For example, hydrochloric acid (HCl) completely dissociates into hydrogen and chloride ions and is highly acidic, whereas the acids in tomato juice or vinegar do non completely dissociate and are considered weak acids. Conversely, strong bases are those substances that readily donate OH– or take up hydrogen ions. Sodium hydroxide (NaOH) and many household cleaners are highly alkaline metal and give upward OH– rapidly when placed in water, the

reby raising the pH. An case of a weak basic solution is seawater, which has a pH near 8.0, close enough to neutral pH that marine organisms adapted to this saline environment are able to thrive in it.
The pH scale ranges from 0 to 14 (Figure 2.17). Anything below vii.0 is acidic, and anything above 7.0 is basic, or alkaline. Since the pH calibration is a negative logarithmic scale, a ten-fold alter in [H+] results in a change of one in pH in the opposite direction. For example, increasing the [H+] from 1 x 10-4 to one x 10-3 decreases the pH from pH 4 to pH 3.
So how can organisms that require a most-neutral pH ingest acidic and basic substances (a man drinking orange juice, for instance) and survive? Buffers are the key. Buffers readily absorb excess H+ or OH–, keeping the pH of the body in homeostasis. For instance, the buffer maintaining the pH of human being blood is a mixture of carbonic acid (H2CO3), bicarbonate ion (HCO–), and carbon dioxide (COtwo). When bicarbonate ions combine with gratuitous hydrogen ions and get carbonic acid, hydrogen ions are removed, moderating pH increases. Excess carbonic acid can be converted to carbon dioxide gas and exhaled through the lungs. This prevents likewise many complimentary hydrogen ions from building up in the claret and dangerously reducing the blood's pH. Conversely, if besides much OH– is introduced into the system, carbonic acid will combine with it to create bicarbonate, lowering the pH. Without this buffer system, the body's pH would fluctuate enough to put survival in jeopardy (Figure 2. 18).
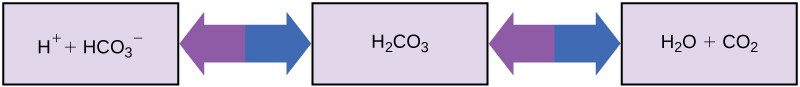
Other examples of buffers are antacids used to combat excess stomach acrid. Many of these over-the-counter medications work in the same fashion equally claret buffers, unremarkably with at to the lowest degree one ion capable of arresting hydrogen and moderating pH, bringing relief to those that suffer "heartburn" after eating.
2.4 | Energy and Life
Past the end of this department, you will be able to:
- Explain the concept of metabolism.
- Explain the difference between potential and kinetic energy.
- Explain how the first 2 laws of thermodynamics relate to living organisms.
2.4.1 Metabolism
Nearly every job performed by living organisms requires energy. In fact, the living cells of every organism constantly utilise free energy. Just equally energy is required to both build and annihilate a building, energy is required for both the synthesis and breakdown of molecules. Other cellular process that require energy include send of signaling molecules, such as hormones and neurotransmitters; ingesting and breaking downwards pathogens, such equally bacteria and viruses; importing nutrients and exporting waste; and many others.
The cellular processes listed higher up require a steady supply of free energy. From where, and in what form, does this energy come? How do living cells obtain free energy, and how exercise they use it? This section volition discuss dissimilar forms of energy and the physical laws that govern energy transfer.
Cellular processes such as building and breaking downward circuitous molecules occur through series of chemical reactions. All of the chemic reactions that take place inside cells, including those that use free energy and those that release energy, are the cell's metabolism. Chemical reactions that require energy to synthesize complex molecules from simpler ones are chosen anabolic reactions, and chemical reactions that release energy as complex molecules are broken down are chosen catabolic reactions.
2.4.two. Potential vs. Kinetic Energy
Energy is defined equally the capacity to do piece of work. When an object is in motion, there is free energy associated with that object because moving objects are capable of enacting a change, or doing work. Think of a wrecking ball. Fifty-fifty a tedious-moving wrecking ball tin do a nifty deal of harm to other objects. Withal, a wrecking ball that is non in motion is incapable of performing work. Energy associated with objects in movement is chosen kinetic energy. A speeding bullet, a walking person, the rapid movement of molecules in the air, and electromagnetic radiations all accept kinetic energy.
At present what if that aforementioned motionless wrecking ball is lifted 2 stories above a car with a crane? If the suspended wrecking ball is unmoving, is there energy associated with it? The answer is yes. The suspended wrecking ball has energy that results from the fact that there is the potential for the wrecking ball to exercise work. This type of energy is called potential free energy. Some other example of potential energy is the energy of water held behind a dam (Figure two . 19).

Potential energy is non simply associated with the location of affair (such every bit a wrecking ball existence held up), just also with the structure of affair. A jump on the footing has potential energy if information technology is compressed; and so does a rubber band that is pulled taut.
Living cells rely heavily on structural potential energy. On a chemical level, the bonds that hold the atoms of molecules together have potential energy. The fact that free energy can be released by the breakdown of certain chemical bonds implies that those bonds have potential energy. In fact, at that place is potential energy stored within the bonds of all the nutrient molecules nosotros eat, considering these bonds can release free energy when broken. This type of potential energy is called chemic energy (Figure 2.20). Chemic energy provides cells with energy past breaking the molecular bonds inside fuel molecules.

two.4.iii The Laws of Thermodynamics
Thermodynamics refers to the study of energy and energy transfer. The laws of thermodynamics govern the transfer of energy in and among all systems in the universe. The first and 2nd laws of thermodynamics are relevant to biological systems and how they convert and exchange free energy with their surroundings.
The Get-go Police force of Thermodynamics
The kickoff law of thermodynamics states that the total amount of energy in the universe is abiding. In other words, energy cannot be created or destroyed. However, energy may be transferred from one class to another. Transfers and transformations of energy take place around united states all the time. Light bulbs transform electrical energy into low-cal energy. Gas stoves transform chemical energy from natural gas into heat free energy. Plants catechumen energy of sunlight into chemical energy stored inside organic molecules. Some examples of energy transformations are shown in Figure 2.21.

The challenge for all living organisms is to obtain energy from their surroundings in forms that they can transfer or transform into usable free energy to exercise work. Living cells have evolved to meet this claiming very well. Chemical energy stored inside organic molecules such as sugars and fats is transformed through a series of cellular chemical reactions into energy inside molecules of ATP. Energy in ATP molecules is easily accessible to practice piece of work.
The Second Law of Thermodynamics
The 2d law of thermodynamics states that the disorder, or entropy, in the universe is ever increasing. None of the energy transfers and transformations in the universe is completely efficient. In every free energy transfer, some corporeality of energy is lost in a course that is unusable. The more energy that is lost by a arrangement to its surroundings, the less ordered and more random the organisation is.
In well-nigh cases, the energy is lost in the form of heat free energy. Thermodynamically, heat energy is the energy transferred from one system to another that is non doing work. For instance, when an airplane flies through the air, some of the energy of the flight plane is lost every bit rut free energy due to friction with the surrounding air. This friction really heats the air by temporarily increasing the speed of air molecules. Likewise, during cellular metabolic reactions, some energy is lost as rut energy. (This is good for warm-blooded creatures like us, because heat energy helps to maintain our body temperature.) In another case, equally molecules at a high concentration in one identify diffuse and spread out, entropy increases (Figure 2 . 22).
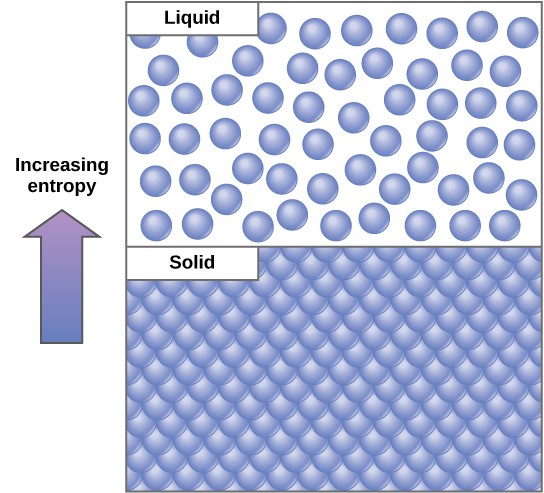
Since living things are highly ordered, they require a constant input of free energy. Essentially, living things are in a continuous uphill battle against this abiding increase in universal entropy.
two.5 | Carbon
By the end of this section, y'all will be able to:
- Explain why carbon is important for life.
- Describe the office of functional groups in biological molecules.
Cells are made of many complex organic (carbon-containing) molecules, such as proteins and carbohydrates, which are especially important for life. The fundamental component of all of these macromolecules is carbon. Carbon atoms tin can form covalent bonds with upward to four dissimilar atoms, making them platonic to form the "backbone" of macromolecules.
2.5.1 Hydrocarbons
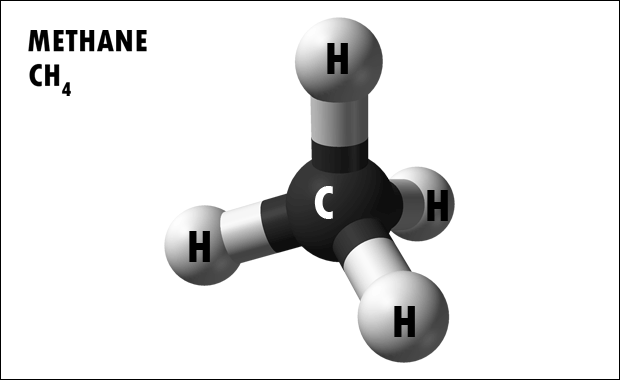
Hydrocarbons are organic molecules consisting entirely of carbon and hydrogen. We often use hydrocarbons as fuels—like the propane in a gas grill or the butane in a lighter. The covalent bonds betwixt the atoms in hydrocarbons store a great amount of energy, which is released when these molecules are burned (oxidized). Methane, an excellent fuel, is the simplest hydrocarbon molecule ( Figure 2. 23 ). Hydrocarbons may exist as linear carbon chains, carbon rings, or combinations of both. Furthermore, individual carbon-to- carbon bonds may be single, double, or triple covalent bonds.
two.v.2 Functional Groups
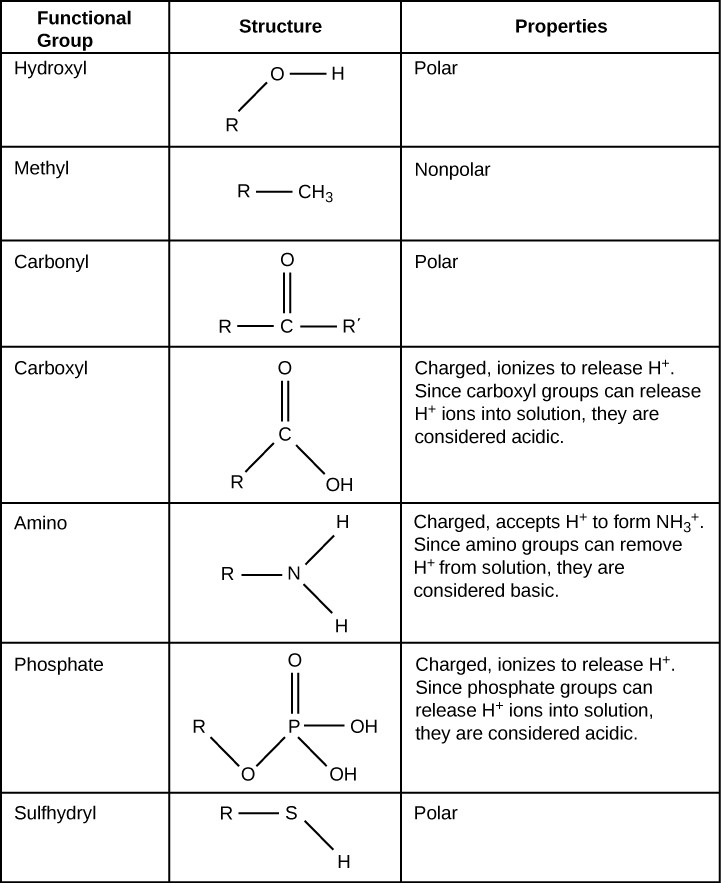
Functional groups are groups of atoms that occur commonly within molecules and confer specific chemic backdrop to those molecules. They are found attached to the carbon "backbone" of macromolecules. Each of the four types of macromolecules—proteins, lipids, carbohydrates, and nucleic acids—has its own feature set of functional groups that contributes greatly to its differing chemical properties and its function in living organisms.
Some of the important functional groups in biological molecules are shown in Figure 2.24. Functional groups are oftentimes classified as polar or non-polar, since that determines whether they are hydrophobic or hydrophilic. For instance, not-polar methyl groups are hydrophobic and polar hydroxyl groups are hydrophilic. Functional groups tin also exist classified as acidic or basic, depending on whether they release or take H+ in solution. Releasing H+ results in a negatively charged functional group, such as phosphate groups or carboxyl groups. Accepting H+ results in a positively charged functional grouping, such as amino groups.
The Chemical Context Of Life,
Source: https://rwu.pressbooks.pub/bio103/chapter/the-chemical-context-of-life/
Posted by: allisonwonelf53.blogspot.com


0 Response to "The Chemical Context Of Life"
Post a Comment